GENMAB (GMAB)·Q4 2025 Earnings Summary
Genmab Beats Q4, Stock Falls 2% as Investors Weigh Pipeline Risks
February 17, 2026 · by Fintool AI Agent
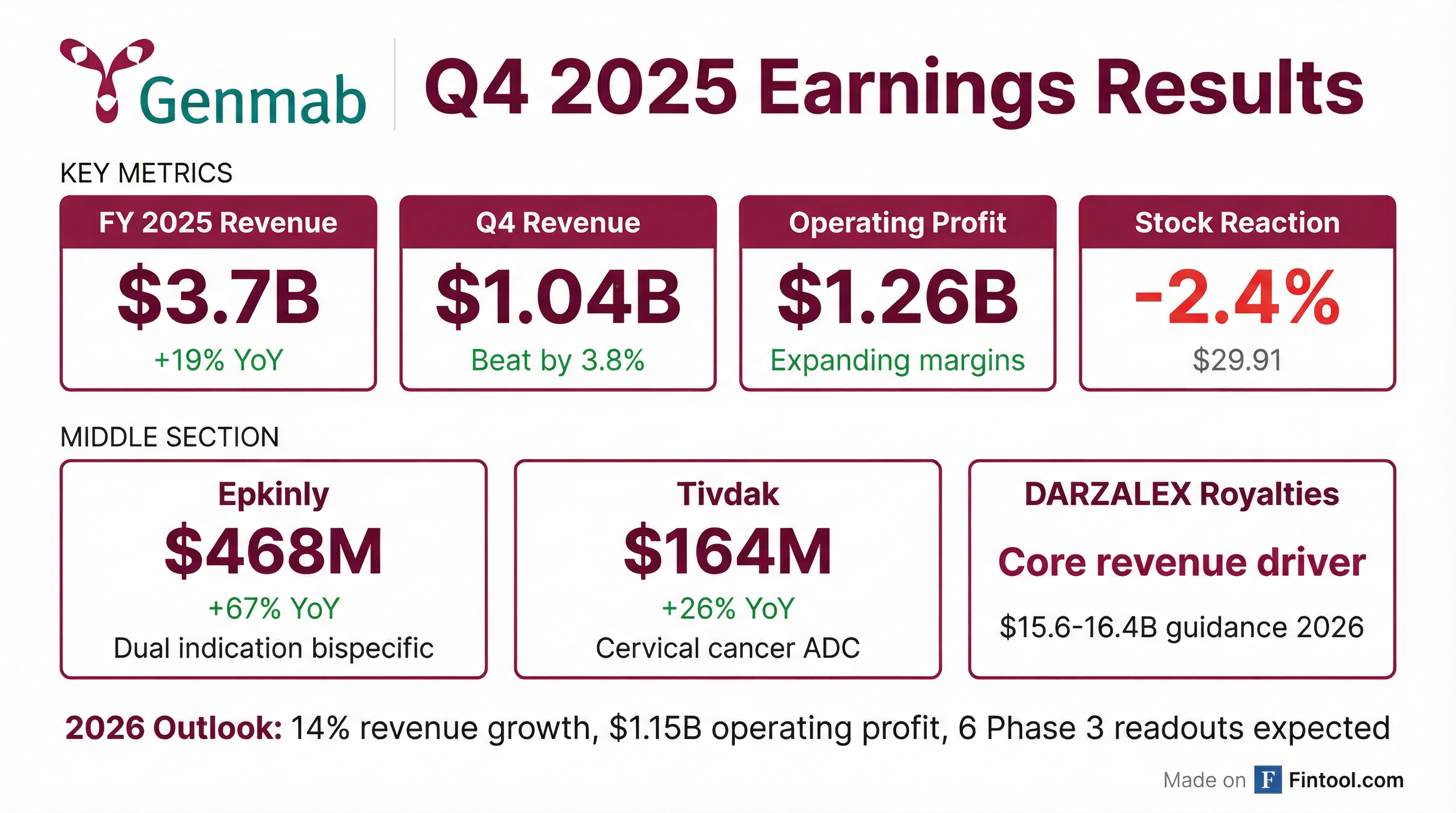
Genmab delivered a solid Q4 2025 with revenue of $1.04B (beating consensus by 3.8%) and EPS of $0.65 (beating by 42%), capping a transformative year that saw 19% revenue growth to $3.7B and operating profit expansion to $1.26B . Despite the beat, shares fell 2.4% to $29.91 as investors digested mixed signals from the Epkinly monotherapy Phase 3 trial and guidance for lower 2026 operating profit ($1.15B) driven by pipeline investments .
Did Genmab Beat Earnings?
Yes — Genmab beat on both revenue and EPS for Q4 2025:
*Values retrieved from S&P Global
For the full year, Genmab grew total revenue 19% to $3.7 billion, fueled by both the royalty portfolio (primarily DARZALEX) and sales from proprietary medicines . Operating profit expanded to $1.26 billion despite strategic investments in commercialization infrastructure and the Merus acquisition .
Proprietary Medicine Performance
Epkinly closed 2025 with regulatory approvals in more than 65 countries, nearly all featuring the dual indication in diffuse large B-cell lymphoma (DLBCL) and follicular lymphoma (FL) . The November 2025 approval of Epkinly in second-line FL in combination with R² is already driving increased uptake .
What Did Management Guide for 2026?
Management provided 2026 guidance reflecting continued investment in late-stage pipeline assets:
The $110M operating profit decline reflects deliberate reinvestment to advance late-stage development for petosemtamab and Rina-S, plus launch readiness activities for multiple potential 2027 product launches . CFO Anthony Pagano emphasized this is "choosing to reinvest part of the operating leverage now to strengthen future growth drivers" .
Capital allocation priorities remain:
- Accelerate late-stage pipeline and commercialization
- Rapid Merus integration to capture value
- Deleveraging to below 3x gross leverage by end of 2027
What Changed This Quarter?
Epkinly Monotherapy Phase 3: Mixed Results
The biggest news was top-line results from the EPCORE DLBCL-1 Phase 3 trial announced in January 2026 :
Positive:
- First Phase 3 to demonstrate PFS improvement in relapsed/refractory DLBCL with CD3/CD20 bispecific monotherapy
- Improvements in complete response rates, duration of response, and time to next treatment
Concern:
- Overall survival did not reach statistical significance
- Management cited COVID-19 (Omicron wave during enrollment) and increasing availability of novel anti-lymphoma therapies as confounders
Chief Medical Officer Tahamtan Ahmadi was clear that pre-specified analyses for these biases were built into the protocol, and discussions with regulators are ongoing . Critically, this does not change expectations for other Phase 3 trials — the combination studies (with lenalidomide, with R-CHOP) were enrolled after the Omicron wave and have different designs .
Merus Acquisition Complete
Genmab completed the Merus acquisition, adding petosemtamab — an EGFR-LGR5 bispecific with potentially transformative data in head and neck cancer — to the late-stage portfolio . The 63% response rate for petosemtamab + pembrolizumab in first-line head and neck cancer is more than 3x higher than the 19% standard of care .
Debt Financing
Genmab completed a $5.5 billion debt offering to fund the Merus acquisition :
- ~$2.5B fixed rate debt
- ~$3B floating rate (with $1.6B hedged to fixed)
- Weighted average effective interest rate: ~6.6%
How Did the Stock React?
Shares closed down 2.4% at $29.91 on February 17, 2026, despite the earnings beat.
The negative reaction likely reflects:
- Lower 2026 operating profit guidance ($1.15B vs $1.26B in 2025)
- Uncertainty around Epkinly monotherapy OS miss and regulatory path
- Near-term margin compression from pipeline investments
However, the stock is still up 43% over the past year, reflecting the transformative growth in proprietary medicines and the strengthening of the late-stage pipeline.
What Are the Key Catalysts in 2026?
Genmab expects up to 6 potentially registrational data readouts in 2026, setting the stage for multiple product launches in 2027 :
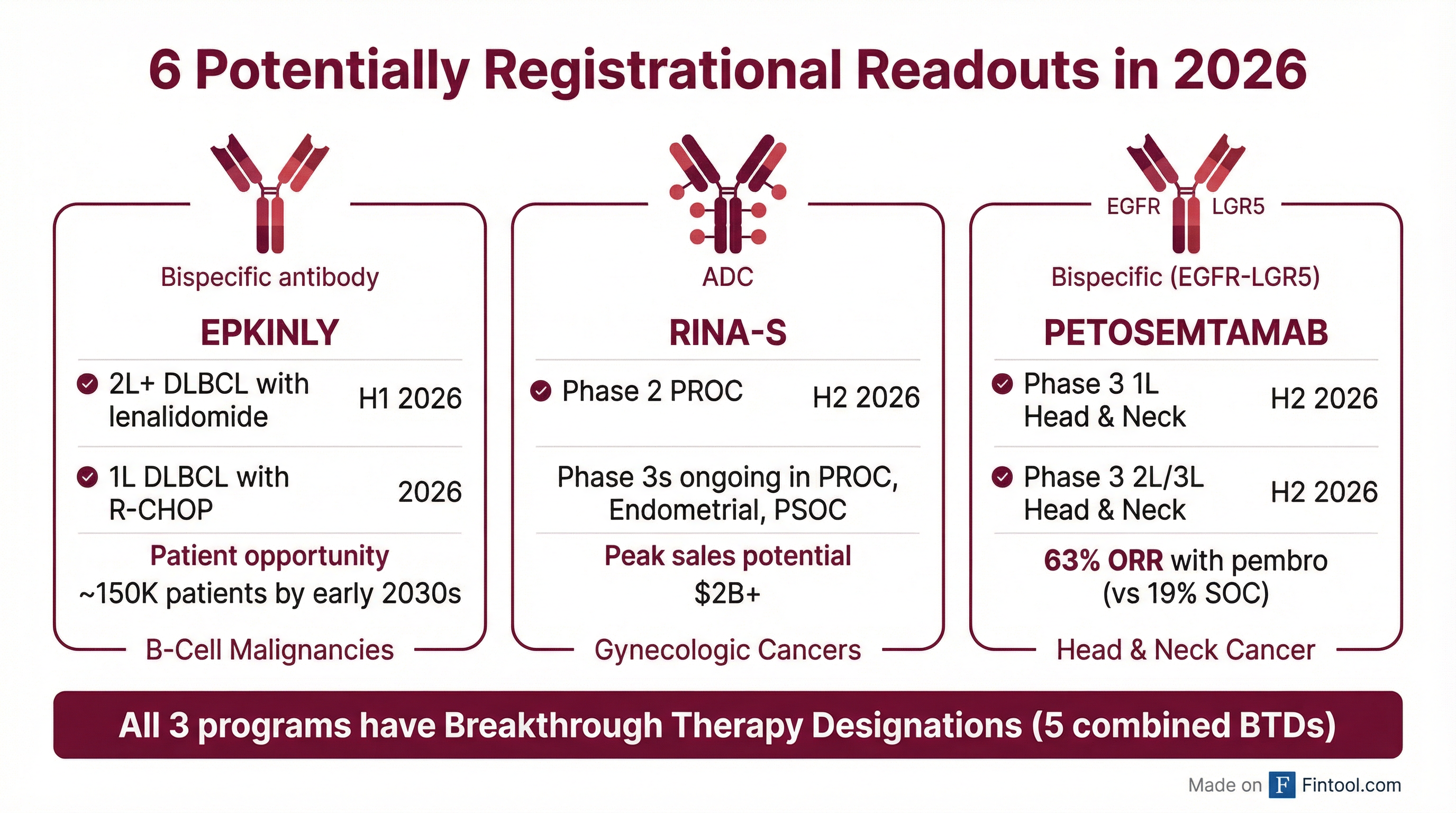
Epkinly (B-Cell Malignancies)
Management expects these trials to move Epkinly into earlier lines of therapy and expand the addressable population from ~27,000 patients today to almost 150,000 by early 2030s .
Rina-S (Gynecologic Cancers)
- Phase 2 PROC readout expected H2 2026 — potentially supportive of accelerated approval
- Peak sales potential: $2B+ across PROC, endometrial, and PSOC
- Designed to broaden eligibility beyond high folate receptor alpha expressers — could expand addressable PROC population by 3x
Petosemtamab (Head & Neck Cancer)
Combined addressable population: ~66,000 patients (41,000 first-line + 25,000 later-line) .
Q&A Highlights
On Epkinly Monotherapy Regulatory Path
"We have, at this point, 3 Phase 3s in diffuse large B-cell... We are extremely comfortable... this is really not a concern on our end in terms of the confirmation trial." — Dr. Tahamtan Ahmadi, CMO
Management emphasized that glofitamab set a precedent, and they have two additional Phase 3 trials reading out in 2026 that could serve as confirmatory studies.
On Petosemtamab Strategy
"The 65% ORR, with 6 CRs, is unprecedented, even in the context of what we know for pembro chemo. We believe that the CDP potentially could be expanded in many different directions." — Judith Klimovsky, CDO
Management is committed to the chemo-free combination (petosemtamab + pembrolizumab) as offering superior value to patients.
On 2026 Operating Profit
"We are choosing to reinvest part of the operating leverage now to strengthen future growth drivers, while continuing to manage costs actively and maintain profitability discipline." — Anthony Pagano, CFO
What Should Investors Watch?
Near-Term (H1 2026):
- Epkinly + lenalidomide Phase 3 data in 2L+ DLBCL
- Regulatory discussions on EPCORE DLBCL-1 confirmatory status
- European/Japan approvals for Epkinly in 2L FL
Back Half 2026:
- Rina-S Phase 2 PROC data — key for accelerated approval path
- One or both petosemtamab Phase 3 readouts in head and neck cancer
- Epkinly + R-CHOP frontline DLBCL data
Execution Risks:
- Integration of Merus organization and pipeline prioritization
- Commercial execution in Europe (new markets)
- Deleveraging progress toward <3x by end of 2027
Data sources: Genmab Q4 2025 earnings call transcript, S&P Global estimates. Stock prices as of market close February 17, 2026.
Related: GMAB Company Profile | Q4 2025 Earnings Transcript | Q3 2025 Earnings